Covid-19
The ChAdOx1 nCoV-19 vaccine was
developed by the University of Oxford and AstraZeneca and manufactured in Serum
Institute of India.
The vaccine works by delivering
the genetic code of the SARS-CoV-2 spike protein to the body’s cells, similarly
to the BNT162b2 vaccine. Once inside the body, the spike protein is produced,
causing the immune...
Ivermectin is an antiparasitic
drug that is approved by the Food and Drug Administration (FDA) for the
treatment of onchocerciasis and strongyloidiasis. Ivermectin is not
FDA-approved for the treatment of any viral infection. In general, the drug is
well tolerated. It is currently being evaluated as a potential treatment for
COVID-19.
Iverm...
The COVID-19 Treatment Guidelines have been developed to
inform clinicians how to care for patients with COVID-19. Because clinical
information about the optimal management of COVID-19 is evolving quickly.
The recommendations in these Guidelines are based on scientific
evidence and expert opinion. Each recommendation includes two...
This guidance document is
intended for clinicians caring for COVID-19 patients during all phases of their
disease (i.e., screening to discharge). This update has been expanded to
meet the needs of front-line clinicians and promotes a multi-disciplinary
approach to care for patients with COVID-19, including those with mild,
moderate, severe, a...
In patients with severe or critical Coronavirus disease 2019 (COVID-19) manifestations, a thromboinflammatory syndrome, with diffuse microvascular thrombosis, is increasingly evident as the final step of pro-inflammatory cytokines storm. Actually, no proven effective therapies for novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) i...
What do we know about this new
SARS-CoV-2 variant?
It’s been snappily named
VUI-202012/01 (the first “Variant Under Investigation” in December 2020) and is
defined by a set of 17 changes or mutations. One of the most significant is an
N501Y mutation in the spike protein that the virus uses to bind to the human
ACE2 receptor. Changes in this...
In a clinical trial of patients with COVID-19, monoclonal antibody were shown to reduce COVID-19-related
hospitalization or emergency room visits in patients at high risk for
disease progression within 28 days after treatment when compared to
placebo. The safety and effectiveness of this investigational therapy
for use in the treatment of C...
FDA Authorizes Monoclonal Antibody for Treatment of COVID-19. The U.S. FDA issued an emergency use authorization (EUA)
for the investigational monoclonal antibody therapy for
the treatment of mild-to-moderate COVID-19 in adult and pediatric
patients.
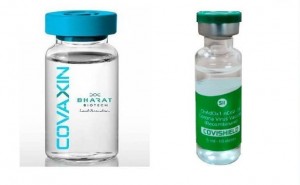
COVAXIN, India's indigenous
COVID-19 vaccine by Bharat Biotech is developed in collaboration with the
Indian Council of Medical Research (ICMR) - National Institute of Virology
(NIV). The indigenous, inactivated vaccine is developed and manufactured in
Bharat Biotech's BSL-3 (Bio-Safety Level 3) high containment facility.
The Drugs Controlle...
India has formally approved the
emergency use of two coronavirus vaccines.
The Drugs Controller General of
India (DCGI) granted its first emergency conditional approvals Jan. 3 for a
pair of COVID-19 vaccines, including Covishield, developed abroad by Astrazeneca plc
and Oxford University and manufactured by the Pune-based S...
The World Health Organization has
granted its first emergency use validation to BioNTech-Pfizer's COVID-19
vaccine.
UN health agency says emergency use
listing ‘opens the door’ for countries to expedite their vaccine approval
processes.
The World Health Organization has
listed Pfizer-BioNTech’s COVID-19 vaccine for emergency use, a crit...
The Oxford University
AstraZeneca coronavirus vaccine has been approved for use in the UK.